
UNE EN ISO 80601-2-61:2019 Medical electrical equipment - Part 2-61: Particular requirements for basic safety and essential performance of pulse oximeter equipment (ISO 80601-2-61:2017, Corrected version 2018-02) - Technické normy

ČSN EN ISO 80601-2-61 (364801) - Zdravotnické elektrické přístroje - Část 2- 61: Zvláštní požadavky na základní bezpečnost a nezbytnou funkčnost přístrojů pro pulzní oxymetrii - říjen 2011 - Technické normy - Ing. Jiří Hrazdil

EN ISO 80601-2-61:2011 - Medical electrical equipment - Part 2-61: Particular requirements for basic

ISO 80601-2-61:2017 Particular requirements for basic safety and essential performance of pulse oximeter equipments - Electrical Safety Testing Laboratory

ISO 80601-2-61:2017 - Medical electrical equipment - Part 2-61: Particular requirements for basic safety and essential performance of pulse oximeter equipment

ISO 80601-2-61 Medical Electrical Equipment - Special Requirements for Basic Safety and Basic Performance of

BS EN ISO 80601-2-61:2019 Medical electrical equipment Particular requirements for basic safety and essential performance of pulse oximeter equipment

DS/EN ISO 80601-2-61:2011 - Medical electrical equipment - Part 2-61: Particular requirements for basic safety and essential performance of pulse oximeter equipment

ONORM EN ISO 80601-2-61:2019 - Medical electrical equipment - Part 2-61: Particular requirements for basic safety and essential performance of pulse oximeter equipment (ISO 80601-2-61:2017, corrected version 2018-02)

ISO 80601-2-61:2017 Particular requirements for basic safety and essential performance of pulse oximeter equipments - Electrical Safety Testing Laboratory

ISO 80601-2-61:2017 Particular requirements for basic safety and essential performance of pulse oximeter equipments - Electrical Safety Testing Laboratory

EN ISO 80601-2-61:2019 - Medical electrical equipment - Part 2-61: Particular requirements for basic

ISO 80601-2-61:2017(en), Medical electrical equipment — Part 2-61: Particular requirements for basic safety and essential performance of pulse oximeter equipment
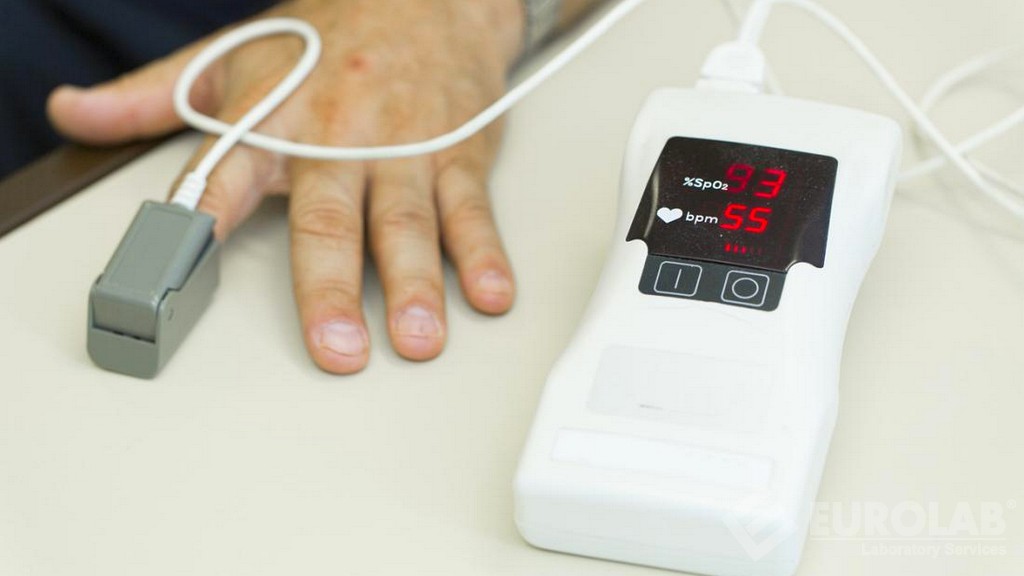
TS EN ISO 80601-2-61 Medicīniskās elektroiekārtas - 2-61.daļa: Pulsa oksimetrijas iekārtu vajadzīgās veiktspējas












